Market Pulse:
Expanding patient access to clinical research
Expanding patient access to clinical research
January 06, 2023 — By Cyndi Bloom

The Market Pulse delivers the latest market trends and issues of interest to those who sponsor, develop, and run clinical trials.
Each issue takes a position on topics like new technologies, trial structures, or regulatory policies related to clinical research. But unlike your typical market insights report, Market Pulse reviews the issues and trends from the person/patient/participant standpoint.
We discuss recent changes in healthcare and clinical research and the ways they can affect the healthcare consumers’ willingness and/or ability to participate in clinical research.
At the beating heart of the clinical trial:
The participant perspective
The participant perspective
Review any ten articles on clinical trials, and you’ll see a huge focus on new technology, new trial structures, and other innovations. Rightfully so. These are transforming how clinical trials are constructed and run. But how often does the literature examine the personal impacts on participants in any depth? Their needs, their issues, their goals?
It’s time to flip the script here: What’s good for the participant is good for you, your trial, your company.
Why would we say this? Because the participant is central to all clinical research. That means before we adopt anything new—like ways of conducting trials or applying technology—we must first evaluate the changes and see if and how they support:
- Broader participant access to research opportunities, including for women and racial and ethnic minorities
- Better participant understanding of research
- More convenient ways to participate
- And ultimately, better participant outcomes
On paper, a new technology, platform, or structure may look good, even great. It might save you time and money. But the true litmus test is whether or not it benefits the participant.

Patient or person? They aren’t mutually exclusive.
Ready to take a trip into the experience of the potential clinical trial participant? Before we dive in, we’d like to make something clear. Those who consider participating in clinical trials aren’t just numbers, aren’t just patients, aren’t even just participants—they’re people. People who lead busy lives with concerns, limitations, and personal issues. And clinical trials will only succeed if they can accommodate these aspects of peoples’ lives.
Let’s get to know someone who’s looking for answers to health problems. Anna, an African American woman in her 60s, suffers from Diffuse Idiopathic Skeletal Hyperostosis (DISH), a rare medical condition that causes bone spurs to develop along the spine and joints. What will she have to deal with to get accepted into an applicable clinical trial?
What am I missing?
It’s not a secret that only 5% of eligible patients participate in clinical research. Why isn’t participation higher? One reason could be that fewer healthcare consumers are hearing about clinical trials from their healthcare providers. In a recent survey of 500 healthcare consumers, we found that 76% said that their doctor had never recommended participation in a clinical study. So much for relying on doctors to refer patients to clinical trials.
Exhibit 1 – Responses to the question: ‘Has a doctor ever recommended participating in a clinical study?’
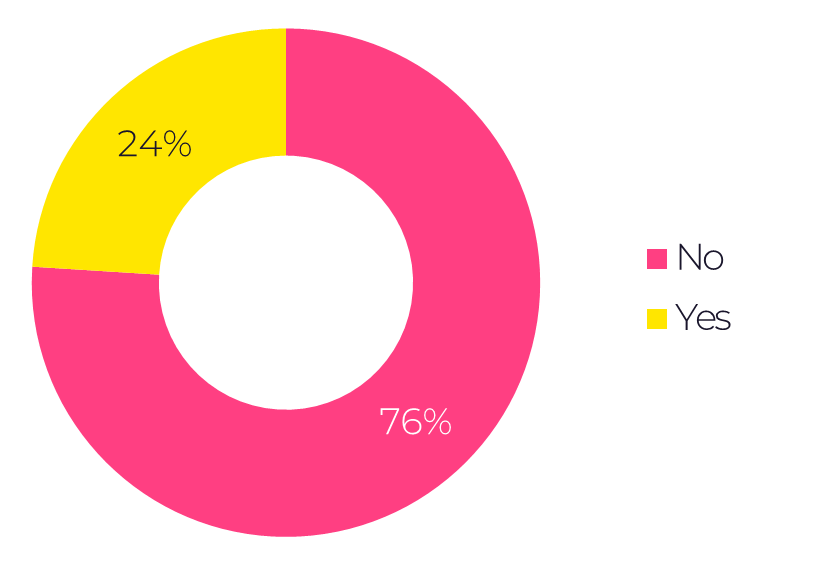
In a survey of 500 healthcare consumers, 76% said that their doctor had never recommended participation in a clinical study.
How about Anna? Her doctor has never once mentioned a clinical trial. So where does that leave her? Like many of those desperate for answers, Anna is looking for information on her own. She’s looking online—putting herself in the driver’s seat for her healthcare journey.

Can I afford it?
Anna is fortunate enough to have learned (with help) about a trial. Luckily, too, her attitudes, knowledge, and beliefs allow her to consider the trial (although some of her relatives don’t feel the same). She’s a good fit for the trial based on the research protocols (she’s gone through pre-qualification online and further qualification by a clinically trained professional). So it’s smooth sailing for Anna, right?
Not exactly. Time or cost or both are top of mind for many potential participants. Especially those who are unemployed or have lower incomes, have inflexible work schedules, or have childcare or family responsibilities (or all three). Anna can relate, as she has a part-time job and cares for her grandchildren two days a week.
So her next concern is her distance from the trial site. Not that much of a problem? Well, did you know that 70% of potential participants live at least two hours away from trial sites? This can put a burden on the participant’s time and wallet due to the need for transportation and lodging.
One study of the financial issues related to an early-phase cancer trial showed that out-of-pocket costs for nearly half of the trial participants ran as high as $1,000 per month. This figure included costs for travel, lodging, missed work, and unexpected medical issues. And while cancer trials may be among the more expensive, patients in trials for other diseases or conditions could face the same types of costs, even if to a lesser degree.
Do I have the physical ability to participate?
People with rare diseases or conditions often have the most difficulty with traveling to a trial site. Either pain and disability hinder travel, or the need to take additional family members with them adds to the cost. Anna’s DISH puts her in considerable pain every day. Getting to a distant site could be just one step too far to travel.
Expanding access to clinical trials
So now that we’ve looked at the challenges Anna and others like her face, let’s look at developments in clinical structure and technology that provide an upside to the participant. Ease the burden on them. Increase the convenience factor. Remove the barriers.
While the developments we’re about to cover may benefit the sponsor—for example, they speed the development of the clinical trial or make it less costly—the true test is if they deliver fundamental benefits to patients. How will they help more people to participate? How will they improve diversity? And ultimately, how will more people see improvements in their quality and longevity of life?
DCTs: Bringing it all back home
The glossary of terms put out by the Decentralized Trials & Research Alliance defines decentralized clinical trials (DCTs) as “A clinical trial utilizing technology, processes, and/or services that create the opportunity to reduce or eliminate the need for participants to physically visit a traditional research site.”
This bears repeating: DCTs reduce or eliminate the need for participants to physically go to a traditional research site. So what does this mean?
By bringing the trial to the participant (at their home and/or nearby clinic), DCTs remove the big geographical blockers and open up the size and inclusivity of the participant pool.
By bringing the trial to the participant, DCTs remove the big geographical blockers and open up the size and inclusivity of the participant pool.
Who’s in? People who are reluctant to spend time in hospital settings, have family commitments, or deal with inflexible work or school schedules. They’re in and that’s all good. But the real bonus is that decentralized trials give more opportunity to women, racial and ethnic minorities, and those with rare diseases to become a part of clinical research. People like Anna. Their participation helps make sure that the medical solutions being developed and tested work as intended—not just for a select few, but for the broadest possible range of people.
Are DCTs the be-all and end-all of clinical research?
Short answer: No.
There are some tests and procedures that simply must be done in a clinical setting. But the use of DCTs is growing for many, many research opportunities. This means decentralized clinical trials can and should be considered where feasible.
Just a caveat here: Not all DCTs are completely virtual, with all aspects of the research conducted from home relying solely on digital reporting and monitoring technologies. So-called hybrid trials may include visits from nurses or require the participants to make one or two visits to a nearby clinic. DCTs may be constructed differently, but that’s OK, as long as they are meeting the needs of the participants.
Have DCTs proven themselves?
DCTs began mostly as a way to continue clinical research during the Covid pandemic. Since then, DCTs have grown legs and taken off. While research on DCTs is somewhat limited to date, it’s increasing.
So what do we know? What are healthcare consumers saying about their interest in clinical trials—especially decentralized trials that use remote technologies? In a recent survey of 1,183 cancer patients and survivors, they shared that only 18% had participated in a cancer clinical trial. But 77% were interested in joining a trial if it was located as close as their regular healthcare provider. Older patients (55 years and older) were much more reluctant to join trials if they had to travel farther than their provider’s office. So in the healthcare consumers’ minds, trials are good. Travel, not so much.
What really stands out in this survey? When questioned about using remote methods and technologies, 80+% said they would happily agree to using remote options, such as “oral medications delivered and taken at home, providing informed consent electronically, and using apps or wearables.”
We’ve seen good results in our own support of decentralized trials, with a trial of a new in-home test for colon cancer. The participants were able to perform and submit the test from home, greatly increasing the pool of participants. While they did have to receive a colonoscopy for comparison to the test results, they could do so at nearby providers. A huge benefit was that more people who needed to be tested were able to do so.
83% of potential patients did not have a colonoscopy previously scheduled before they became involved in the qualification process.
This important trial made it possible for a new product to be developed that helps catch colorectal cancer at an early stage and helps extend the lives of those diagnosed with the disease.
There are growing pains
So what needs to be done to ensure DCTs deliver benefits for participants?
A quick look into research into DCTs reveals a few issues, some with answers and some still unsolved. They range from participant knowledge, to privacy, to safety issues. Let’s take a look.
What’s impacting Anna’s experience the most? Technology literacy (or lack of it). DCTs extensively use remote technologies. But not everyone understands how to operate them. This lack of tech knowhow could put people—and outcomes—at risk.
DCTs extensively use remote technologies. But not everyone understands how to operate them. This lack of tech knowhow could put people—and outcomes—at risk.
Although 85% of individuals owned smartphones in the United States as of 2020, far fewer people 65+ do. What to do?
- Provide tablets for those who don’t have smartphones.
- Educate empathetically (something 83bar does every day). Bring these people up to speed and help them feel comfortable with technology.
Anna has the drive to participate, but not the full knowledge of how to use reporting technology. Tutorials and phone support get her on the right track and help her feel more confident.
Other issues that may apply to minorities or people with lower incomes (like Anna) concern privacy and even safety.
- In some cultures, home nurse visits can cause issues for participants. Community members are sometimes suspicious if new nurses visit each time and come from different races or ethnicities than participants. So how do we fix this? By ensuring the home health nurse is the same one at every visit and that the nurse shares the patient’s race and/or ethnicity.
- Expensive equipment and/or drugs delivered to a home in a low-income neighborhood can bring unwanted attention—and even put participants at risk of break-ins.
Another way of coming at these issues is through use of mobile facilities. They can be located near patients and come with the staff and necessary equipment. This means that:
- Visits don’t have to be made to the home (so no fun for prying neighbors)
- Equipment doesn’t have to be stored in the participant’s home (so less chance of robbers visiting)
With the crime rate on the high side in Anna’s neighborhood, she likes the option of a mobile site. This way she doesn’t have to keep expensive equipment and drugs in her home.
We’ve been asking what it takes to boost participation in clinical research and suggesting answers where we can. But we believe it’s important to take the focus on the participant and their needs one step farther. How better to do that than by asking for input from patients and advocacy groups? This means giving voice to those who have been left out of traditional trials in the past—whether they’re low income, minorities, or people with rare diseases.
We believe it’s important to take the focus on the participant and their needs one step farther by asking for input from patients and advocacy groups.
Why not reach out to organizations like PCORI, the Patient-Centered Outcomes Research Institute? PCORI has supported more than 122 clinical effectiveness research studies and related projects. PCORI is looking to help patients at risk for disparities and their caregivers make better-informed decisions about improving their health. Want to gain some insights on patient engagement? Read patient reflections on studies funded by PCORI to engage diverse communities in asthma care.
Where do we go from here?
Let’s help Anna and people like her to improve research into products and treatments that deliver better outcomes for everyone. DCTs hold promise for opening up access to all who want to take part in clinical trials to help themselves or to help others.
You’re invited. Join us in focusing on the participant.
In searching for ways to broaden access to research and improve the experience for participants like Anna. We’d love to hear what you have to say. Visit our LinkedIn page to see more about this topic and leave your feedback.




